|
11/15/2022 0 Comments Rutherford experiment conclusion 
According to Thomson's model, the positive charge is uniformly spread throughout the atom, which creates a weak electric field. The experiment results obtained from the alpha particles scattering experiment contradict Thomson's model. Thomson's atomic model Contrast between the experiment and Thomson's model According to him, the positive charge was uniform throughout the atom, and the magnitude of it was equal to negative charges. He explained an atom as a sea of positive charge with floating negatively charged electrons. In the 1900s, Thomson's atomic model was dominant. Most particles which are scattered have less than 90° deflection while few particles cross 90°.īefore proceeding to analyse the outcomes of the experiment, let's quickly review Thomson's atomic model. they continue to travel a straight path as if the gold foil was absent. The particles leaving the foil after the strike make tiny glowing flashes on the screen called scintillations, which can be measured using a microscope.Īs we can see from the figure, most particles remain undeflected, i.e. A circular zinc sulphide fluorescent screen surrounds the foil. 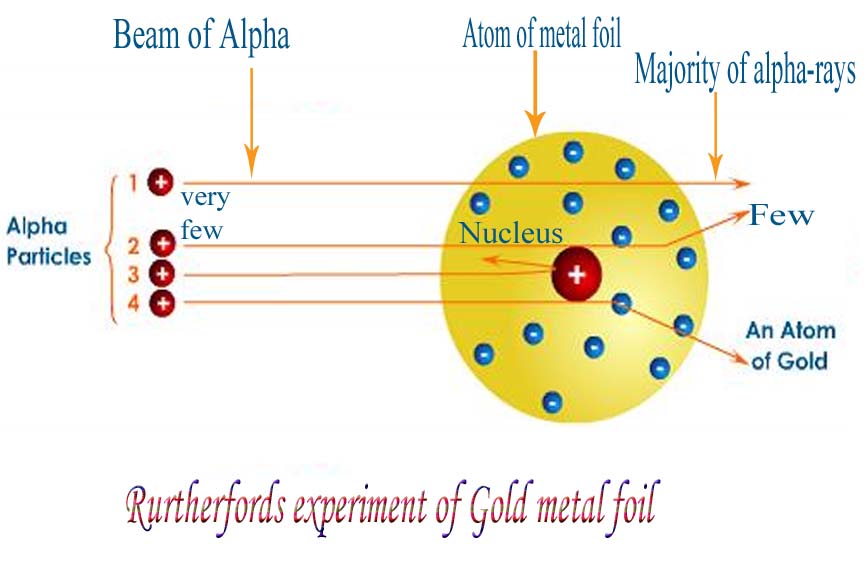
The beam moves through space and strikes the gold foil of a few thousand atoms thick. The lead block acts as a shield and sharpens the beam. The illustration above depicts a radioactive source enclosed in a lead block liberates alpha particles. 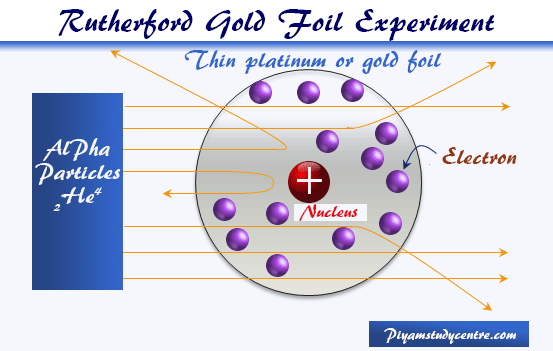
When Rutherford along with his colleague shot alpha particles, the positively charged helium nuclei, on a very thin gold foil, unexpected scattering of the particles was observed. All the experiments can be summarized using the illustration below. The experiment was not a single experiment but a series of experiments. The outcomes of the experiments debunked the idea the positive charge was uniformly distributed in the atom and demonstrated the presence of a central nucleus, where all of its positive charge is concentrated. The experiments led to the discovery of the nucleus and gave birth to the nuclear model of the atom. 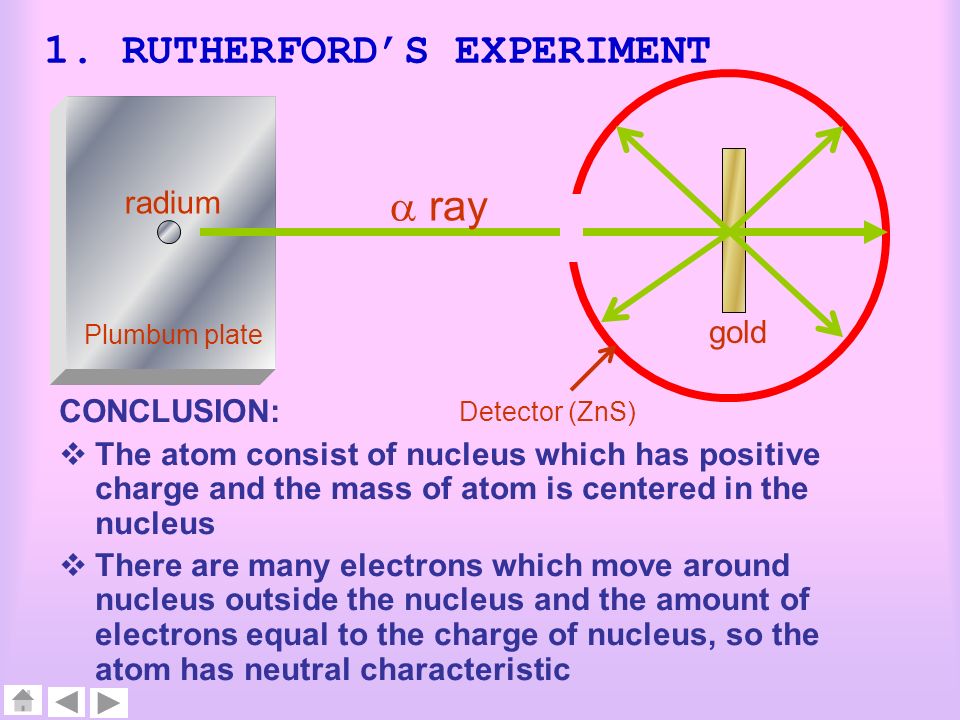
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
